State of the Art in Hairy cell leukaemia (‘Hairy cell leukaemia ('HCL')’) Targeted Therapies: Q1 2026 Technical Report
Hi everyone,
I approach Hairy cell leukaemia (‘Hairy cell leukaemia ('HCL')’) from an engineering mindset, tracking biotechnology trends and clinical trial data to understand the mechanics of upcoming treatments.
I have compiled a brief technical report on the latest advancements (as of mid-February 2026) regarding targeted therapies—specifically cellular engineering like CAR T-cell therapy (‘CAR T-cell therapy ('CAR-T')’) and novel NK cell engagers.
I am sharing this as a test run. If you find this type of technical summary useful, please let me know in the comments. If there’s interest, I can post similar updates periodically, but only when there is significant new information, to ensure the content remains fresh and valuable without being repetitive.
DISCLAIMER: This post is for informational and educational purposes only, based on publicly available data. It is intended to highlight current biotech trends from a technical perspective. It is NOT medical advice. I am not a doctor. Always consult your hematologist or oncologist before making any treatment decisions.
TECHNICAL REPORT: ADVANCES IN HEMATO-ONCOLOGY (Hairy cell leukaemia (‘Hairy cell leukaemia ('HCL')’) & TARGETED THERAPIES)
Review Date: February 14, 2026 Focus: Cellular engineering & Bispecific/NK Engagers
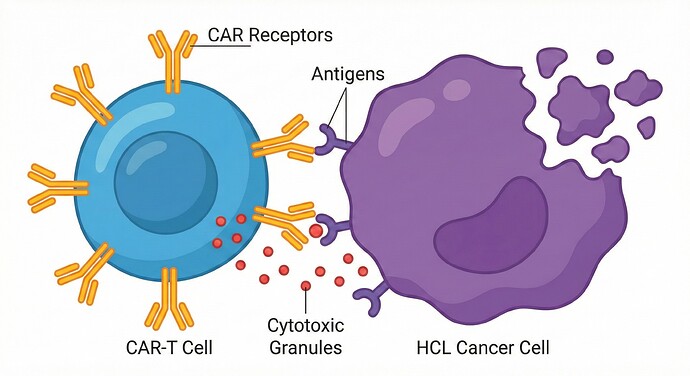
1. Anti-CD22 CAR T-cell therapy (‘CAR T-cell therapy ('CAR-T')’) Therapy (NCI, Kite, Novartis)
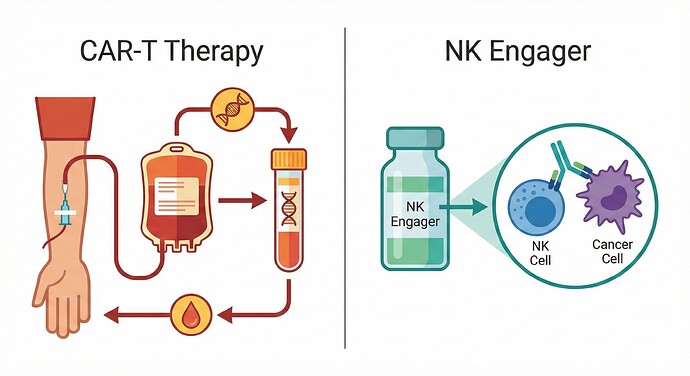
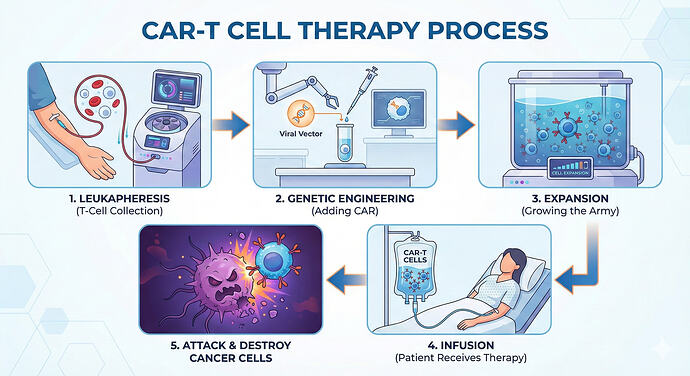
CAR T-cell therapy (‘CAR T-cell therapy ('CAR-T')’) technology in the context of Hairy cell leukaemia (‘Hairy cell leukaemia ('HCL')’) is evolving toward consolidation therapy aimed at preventing relapses after standard treatments.
-
NCI (National Cancer Institute): * New Trial (NCT07328503): Announced in early February 2026, the NCI is investigating the use of CD22 CAR T-cell therapy (‘CAR T-cell therapy ('CAR-T')’) cells as a consolidation extension for patients who have previously received CD19 targeted therapy. The goal is to close the loop on remission and eliminate the risk of “antigen escape.”
- Hairy cell leukaemia (‘Hairy cell leukaemia ('HCL')’) Specifics: The Phase I trial (NCT02315612) dedicated to Hairy cell leukaemia (‘Hairy cell leukaemia ('HCL')’) and Hairy cell leukaemia (‘Hairy cell leukaemia ('HCL')’)-variant remains active at NIH Bethesda. Recent data confirms the extremely high density of CD22 on hairy cells, making it a highly stable and precise target.
-
Kite (Gilead) & Novartis: * Focus is shifting toward bicistronic platforms (CD19/CD22) to provide targeting redundancy. Additionally, reports indicate a push toward minimizing manufacturing time (vein-to-vein time) to under 7 days using platforms like Novartis’s T-Charge.
2. Innate Pharma (IPH) – ANKET® Platform & CD123 (IPH6101)
3. AstraZeneca – The Post-Moxetumomab Pipeline
4. Next Generation: CAR-HSC Architecture (Modifying the “Factory”)
While the classic CAR T-cell therapy (‘CAR T-cell therapy ('CAR-T')’) process relies on modifying mature lymphocytes, the latest data introduces a fundamental shift in treatment architecture. Genetic engineering now transfers the modification vector directly to the level of Hematopoietic Stem Cells (HSCs). This solves the hardware-level problem of the limited lifespan inherent in classic cellular therapies.
Limitations of Classic CAR T-cell therapy (‘CAR T-cell therapy ('CAR-T')’) (Modifying Endpoint Units): The current protocol deploys a finite pool of reprogrammed T-cells. From a technical standpoint, this system has a predetermined lifespan. Over time, these cells undergo T-cell exhaustion; their division cycle loops and eventually shuts down. In the case of resistant cells (e.g., in Hairy cell leukaemia (‘Hairy cell leukaemia ('HCL')’)), this functional decline opens a pathway for relapse.
The CAR-HSC Model (Source Modification): In this approach, a viral vector carrying the CAR receptor code is introduced directly into the patient’s stem cells within the bone marrow. The modification targets the core of the hematopoietic system—the “factory”—rather than just the individual “soldiers.”
-
System Efficiency (Self-Renewal): The engineered HSCs acquire the capacity for continuous, lifelong production of new, functional units.
-
Systemic Effect (Full Spectrum): The upgraded “factory” releases a complete spectrum of armed immune forces into the bloodstream. This includes not only targeted CAR T-cell therapy (‘CAR T-cell therapy ('CAR-T')’) lymphocytes but also CAR-NK (Natural Killer) cells and CAR-Macrophages. The immune system receives a permanent update at the source level.
-